
Researchers at UHN’s KITE Research Institute have recently leveraged the power of deep learning to improve nerve signalling interpretation, paving the way for more effective neuroprosthetic devices and offering new hope for those with impaired motor function.
Neurological injuries and amputation frequently result in reduced motor function and diminished quality of life. Neuroprosthetics are small, implanted devices designed to help restore lost movement or sensory function by using electrical impulses to stimulate nerves—a process known as neurostimulation.
These devices use small electrodes to capture sensory information from the nerves and the environment to refine movements. However, interpreting these nerve signals is challenging due to the nature of the electrodes. Placing electrodes inside the nerve provides clearer signals but poses a risk of neural tissue damage. Electrodes positioned outside the nerves are less invasive but yield lower signal quality.
Dr. José Zariffa, Senior Scientist at the KITE Research Institute and senior author of the study, led a team that applied deep-learning models to enhance nerve signal interpretation.
“Deep learning is a form of artificial intelligence that excels at analyzing complex data and recognizing patterns,” explains Dr. Zariffa. “By applying these models, we can decipher complex and low-quality nerve signals and extract more precise information.’
To evaluate the effectiveness of these models, the research team tested different deep-learning models on nerve signals collected from the sciatic nerve—one of the largest nerves in the body running from the lower back to the foot. They analyzed the model’s ability to understand the nerve signals and classify them based on where they originated in the body.
“We found that deep learning significantly improved nerve signal interpretation accuracy,” states Aseem Partap Singh Gill, previous undergraduate student in Dr. Zariffa’s lab and first author of the study. “The best-performing model achieved an accuracy of around 93%, demonstrating how deep learning can enhance nerve signalling interpretation and improve neuroprosthetics for individuals living with disabilities.”
Future research aims to apply these models to analyze signals from multiple electrodes, different nerves, and more complex movements.
This work was supported by the Natural Sciences and Engineering Research Council of Canada and UHN Foundation. Dr. José Zariffa is an Associate Professor in the Institute of Biomedical Engineering and is cross-appointed at the Edward S. Rogers Sr. Department of Electrical & Computer Engineering and the Rehabilitation Sciences Institute at the University of Toronto.
Gill APS, Zariffa J. Time series classification of multi-channel nerve cuff recordings using deep learning. PLoS One. 2024 Mar 12. doi: 10.1371/journal.pone.0299271.
Researchers at UHN's Princess Margaret Cancer Centre have discovered a promising approach to boost the effectiveness of immunotherapy cancer treatments by manipulating the metabolism of a specific type of immune cell.
Cytotoxic T cells are a type of white blood cells that play a key role in the surveillance and elimination of abnormal cells, including cancerous ones.
Some tumours can evade T cell recognition and attack, which can lead to disease progression. One common strategy in cancer immunotherapy is Adoptive Cell Therapy (ACT), which involves using a patient's own immune cells to fight cancer.
In ACT, immune cells such as T cells are collected from the patient's blood or tumour tissue, modified or activated to enhance their ability to fight tumours, and then infused back into the patient.
"Although successful in some cases, cell therapy typically doesn't offer long-lasting benefits for most patients," explains Dr. Pamela Ohashi, Senior Scientist at Princess Margaret Cancer Centre and senior author of this study. "We aim to improve the way ACT is made to increase its effectiveness long-term."
Previous research indicates that directing interventions towards metabolic stress pathways, which have been conserved through evolution, like nutrient deprivation or energy production, can enhance the ability of T cells to control tumours.
The team set out to study an important molecule that cells produced in response to nutrient starvation, called GCN2 (kinase general control non-depressible 2).
"We knew that when GCN2 is activated, it generally reduces overall protein production, leading to the expression of molecules that coordinate various cellular activities, like protein uptake," explains Dr. Michael St. Paul, Post-Doctoral researcher at Princess Margaret Hospital and first author of this study. "However, the specific role of GCN2 in cytotoxic T cells remains unclear."
To study this, researchers started by investigating amino acid depletion, specifically the lack of arginine, which is recognized for its crucial role in immune cell function.
By moving activated cytotoxic T cells from standard medium to one depleted of arginine, scientists noted an increased activation of GCN2-induced stress response. This was shown by an increase in the production of immune-regulating molecules (cytokines) and enhanced energy production through oxidative metabolism.
To validate these results, they used halofuginone (halo), a GCN2 activator, and observed comparable impacts on T cell function and oxidative metabolism.
"Interestingly, this metabolic shift persisted even after removing the halo from the cells, indicating a sustained change in the metabolism of T cells," explains Dr. Sam Saibil, Staff Oncologist at the Princess Margaret Cancer Centre. "This means that harnessing the intrinsic capabilities of the immune system could lead to more targeted and durable therapeutic interventions."
This study helped understand how GCN2 signaling affects T cell responses to arginine levels, shedding light on potential therapeutic strategies for enhancing immune responses against cancer.
"Our research opens doors to enhancing various forms of cancer immunotherapy by targeting GCN2, signaling a potential paradigm shift in cancer treatment strategies," concludes Dr. Ohashi.
This work was supported by the Canadian Institutes for Health Research Foundation and the Princess Margaret Cancer Foundation.
St Paul M, Saibil SD, Kates M, Han S, Lien SC, Laister RC, Hezaveh K, Kloetgen A, Penny S, Guo T, Garcia-Batres C, Smith LK, Chung DC, Elford AR, Sayad A, Pinto D, Mak TW, Hirano N, McGaha T, Ohashi PS. Ex vivo activation of the GCN2 pathway metabolically reprograms T cells, leading to enhanced adoptive cell therapy. Cell Rep Med. 2024 Mar 19;5(3):101465. doi: 10.1016/j.xcrm.2024.101465.
To bridge education gaps and foster cultural sensitivity, a study from UHN’s The Institute for Education Research (TIER) explored the perceptions of nursing students on the experiences of Indigenous peoples within Canada’s healthcare system. Their results revealed strategies for tailoring nursing education to ensure more culturally safe and compassionate care.
Indigenous communities have long faced health inequities marked by a higher burden of illness, inequitable health factors, and premature mortality. Indigenous peoples encounter discrimination every day, including in healthcare settings.
In 2015, the Truth and Reconciliation Commission of Canada developed 94 calls to action to address the impact of residential schools and promote reconciliation in Canada. This involved urging nursing students to understand Indigenous history and be trained to provide culturally safe care.
The Canadian Association of Schools of Nursing developed strategies to integrate Indigenous health content into nursing education; however, the effectiveness of these educational initiatives remains largely unknown.
Dr. Kateryna Metersky, Affiliate Scientist at TIER and a nurse at UHN’s Toronto Western Hospital, led a research team to explore nursing student knowledge and understanding of the experiences of Indigenous peoples within the Canadian health care system and identify how educators can better equip students.
Fifteen nursing students from a university in Ontario, Canada, discussed and shared reflections on a video from the Aboriginal Peoples Television Network, the first national Indigenous broadcaster in the world, entitled “Indigenous Peoples and the Problems in Health Care”.
In their reflections, students showed a deep understanding of the complex factors impacting Indigenous health, including racism, historical injustices, and structural barriers to accessing healthcare, particularly those living in remote communities.
Students also identified strategies to help provide culturally safe care, such as building trust-based relationships, establishing rapport, eliminating unconscious biases, and developing advocacy skills to challenge social inequities within Canada’s complex healthcare system.
“Examining the educational content of nursing programs is important to assessing the value of education provided and tailoring future curriculum to better address knowledge gaps,” concludes Dr. Metersky. “Future work should be conducted to determine the impact of tailored curriculum on preparing students to become culturally compassionate practitioners.”
This work was supported by UHN Foundation.
Metersky K, Chandrasekaran K, Ezekiel S. Undergraduate Nursing Student Reflections on Indigenous Peoples' Experiences With the Canadian Health Care System. Nurs Educ Perspect. 2024 Mar 7. doi: 10.1097/01.NEP.0000000000001255.
Dr. Lillian Siu was inspired to become an oncologist in her teenage years. After her mother was diagnosed with breast cancer, she accompanied her to hospital visits for twelve years, receiving chemotherapy at Princess Margaret Cancer Centre (PM).
“I felt very comfortable in the environment and it started to feel like home. Then I knew it was my calling to study cancer,” says Lillian, who went on to complete a Doctor of Medicine degree in 1991 and a medical oncology residency in 1995, both at the University of Toronto.
Today, Lillian is a global leader in drug development, and through her long career, she has seen life-changing advances that have revolutionized patient care.
She is also the first Canadian to be named President-Elect for the American Association for Cancer Research – the largest cancer research organization dedicated to accelerating the conquest of cancer.
“It is both daunting and exciting,” says Lillian of this prestigious global role. “I’m excited to learn different aspects of cancer research and foster collaboration with other cancer centres. I think in a world where disparity is a definite issue, it will allow us to break some barriers, ensuring that advancements and resources are accessible.”
Lillian has dedicated her life’s work to early drug development.
When she started her career, there were only chemotherapy drugs. “There was no immunotherapy or antibody drug conjugates. It was at the dawn of the molecular era that empowered drug discoveries.”
Formal training for early phase cancer drug development was only available in a few centres in the world. San Antonio, Texas, one of the hubs with extensive pharmaceutical collaborations, attracted early drug developers to the field.
Lillian wanted to apply for a fellowship there, but the spots had been filled and she needed to wait for another year. Instead of simply waiting around, she sought out to create a similar training experience for herself, despite the absence of such a program.
“I asked my supervisor Dr. Malcolm Moore at that time: ‘I want to go to San Antonio in a year. Can I do a fellowship with you similar to that?’ I was fascinated to learn more about early drug development, and I wanted to be prepared for San Antonio. This would allow me to understand more about early phase trials.”
Lillian completed an oncopharmacology fellowship at PM, before heading south for San Antonio, where she encountered one of the first early drug development studies of EGFR inhibitors and trained with many vanguards of the field. She came back in 1998 and after three years, she became the Director of the Phase I Program and co-Director of the Drug Development Program at PM. Her efforts have brought the two programs to a much larger scale, training over 70 people in the world who aspired to become drug developers and clinical trialists.
“The message is that it's okay to create a new path,” says Lillian, proud of the fact that she was able to be an innovator, “I've always wanted to be the one that can go far by having original ideas.”
“Bringing various expertise together allows us to transform original thinking into actions.” Lillian identifies her role as close to being a catalyst, accelerating translational research by finding different dry lab and wet lab collaborators to work together.
One of the pinnacle examples is the INSPIRE study (NCT02644369), a highly impactful clinical trial that studied the effects of the immunotherapy, pembrolizumab, in patients with advanced solid tumours.
This flagship phase II study was led by Dr. Lillian Siu and Dr. Pamela Ohashi at PM. The two investigators, who are respectively renowned for clinical drug development and T-cell immunology research, were tasked to bring basic, translational, and clinical immunology together. They established the Tumor Immunotherapy Program (TIP) at PM that conducted the study and many more immunotherapy trials.
The INSPIRE study started in 2016, enrolling 106 patients with advanced solid tumours spanning a variety of tumour types including head and neck cancer, breast cancer, ovarian cancer, melanoma, and mixed-type cancer. The team secured pembrolizumab, an immune checkpoint inhibitor from Merck, the pharmaceutical company, to treat patients and evaluate gene changes and immune biomarkers in tumor and in blood in response to the treatment.
One of the key discoveries from INSPIRE published in Nature Cancer showed that early and routine circulating tumour DNA (ctDNA) testing can be used to monitor response to prembrolizumab, providing important evidence for the development and application of liquid biopsy. It has also contributed the most relevant data that led to the funding approval by Medicare for ctDNA monitoring for cancer patients receiving immunotherapy in the U.S.
INSPIRE sets up a ‘gold standard’ for investigator-initiated therapeutic studies. Multiple publications have emerged from data generated from the study. Another achievement from INSPIRE, reported by two co-lead authors Dr. Eric Zhao and Dr. Enrique Sanz-Garcia, showed that methylation and fragmentation status of cell-free DNA in patient plasma samples can predict patient survival (published in Cancer Discovery in 2024). This technique can estimate ctDNA levels without the need to access tumour tissues, which makes it very adaptable for clinical settings.
In addition to these outputs and in collaboration with Dr. Trevor Pugh, the team is currently looking at the T-cell receptors (TCR), collected from patients’ biological samples before and during their treatment. “We hope to understand what changes in the TCR repertoire can predict patients’ response to the therapy, what are the signs to tell which patients will have better outcomes, and how their TCR diversity varies in the blood and the tumour,” says Lillian.
“What’s more important than each discovery is the multidisciplinary thinking fostered by the collaboration. The scientific and clinical teams work together to have mutually beneficial crosstalk, which elevates the discovery to a higher level,” says Lillian.
Building on top of the established collaborations and the discoveries of the INSPIRE study, Lillian initiated the NIP-IT! Program, which includes four clinical studies since its inception, each targeted at a different type of tumour, to intercept and intervene in cancer relapse based on the ctDNA levels in the blood. NIP-IT! provides a platform to include future interception studies in other tumour types. As the name suggests, the ultimate goal of NIP-IT! is to nip cancer in the bud.
Today, Lillian holds many important positions at PM. Not only does she have both clinical and research appointments as a Senior Medical Oncologist and Senior Scientist, but she is also the Director of the Phase I Clinical Trials Program, Co-Director of the Bras and Family Drug Development Program, Clinical Lead for the Tumor Immunotherapy Program and BMO Chair in Precision Genomics at PM.
Lillian enjoys working in a multidisciplinary group. Over the years, collaborating with multiple academic and pharmaceutical teams has earned her high esteem within the international oncology community. She has served on the Board of Directors for the American Society for Clinical Oncology (ASCO) and the American Association for Cancer Research (AACR).
Juggling multiple programs and holding many important roles, Lillian has evolved into an expert multitasker: “I try my best to manage multiple tasks at the same time while keeping my standard to do a decent job." Whether it's managing online meetings, emails, or updating medical records, efficiency is key in her fast-paced world.
"At the end of every day, I make sure I've closed all the loops," she emphasizes showing tenacity and commitment.
"To promise something and not deliver is not in my code of professionalism," she asserts, “because that means I have no way to judge my skills and no way to predict my time.”
On female leadership, Lillian encourages women not to focus on how they are underserved, but to show others what they can do.
“I don't want to ignore the fact that there is a disparity, that there are fewer female researchers or leaders in high positions.”
“But I've never been threatened by that. I never feel that I cannot speak up in a panel of men. And I never feel that their science is better than my science if I consider myself on equal footing.”
“How do you prove to somebody that you're not threatened by it? You do better. It is more important for me to demonstrate what I can do than to promise you what I can do.”
Meet PMResearch is a story series that features Princess Margaret researchers. It showcases the research of world-class scientists, as well as their passions and interests in career and life—from hobbies and avocations to career trajectories and life philosophies. The researchers that we select are relevant to advocacy/awareness initiatives or have recently received awards or published papers. We are also showcasing the diversity of our staff in keeping with UHN themes and priorities.
Amidst the ongoing opioid crisis, the number of individuals with opioid use disorder is on the rise. Opioid use disorder, a chronic, life-limiting illness, leads to substantial impairment and distress. As the population with opioid use disorder ages, there is a growing need for access to palliative care at the end of their lives.
This study led by Dr. Jenny Lau, the medical director of the Harold and Shirley Lederman Palliative Care Centre at Princess Margaret Cancer Centre, delves into the characteristics of people with opioid use disorder at the end of their lives and investigates the accessibility of palliative care for this demographic.
The research, based on a comprehensive study including information from ICES on 679,840 deaths, revealed that 11,200 of the deceased (1.6%) had opioid use disorder. Individuals with opioid use disorder died at younger ages than those without the disorder (50 years v. 78 years) and were more likely to reside in marginalized neighborhoods.
Comparatively, individuals with opioid use disorder were 16% less likely to receive palliative care than those without the disorder, particular in clinics and at home. This disparity is most likely linked to the high number of individuals with opioid use disorder who die suddenly from drug poisoning and do not have an opportunity to receive palliative care. The most common reasons why physicians provided palliative care for individuals with opioid use disorder were cancer, liver cirrhosis, and sepsis.
Dr. Lau, the lead author of this study, highlights, “The majority of conversations about the opioid crisis focus on the high number of opioid toxicity deaths. The unfortunate reality is that people with opioid use disorder are dying young from other causes as well. If we can improve our understanding of this population and the healthcare that they receive at the end of their lives, we can identify opportunities to intervene and improve their quality of life.”
This research underscores the urgency of addressing the disparities in palliative care provision for individuals with opioid use disorder. By understanding these discrepancies, researchers can pave the way for targeted interventions to ensure equitable end-of-life care for all.
With the proper training in both palliative care and addiction medicine and attention to socioeconomic inequalities, healthcare providers can significantly improve the quality of life for patients with opioid use disorder in their final days.

(L-R) Co-principal Investigators: Dr. Jenny Lau, the medical director of the Harold and Shirley Lederman Palliative Care Centre at Princess Margaret Cancer Centre, and Dr. Sarina Isenberg, chair in mixed methods palliative care research at Bruyère Research Institute, and adjunct scientist, ICES, Ottawa, Ontario.
This work was supported by funding from ICES, which is funded by a grant from the Government of Ontario. This work was also supported by the Ontario Health Data Platform (OHDP), a Province of Ontario initiative to support Ontario’s ongoing response to COVID-19 and its related impacts, and The Princess Margaret Cancer Foundation. This study also received funding from the Canadian Institutes of Health Research and Health Canada’s Health Care Policy and Strategies Program. The views expressed herein do not necessarily represent the views of Health Canada and Canadian Institutes of Health Research.
Jenny Lau reports honoraria from the University of Toronto and travel funding from the Canadian Institutes of Health Research and Health Canada. She is a member of the Global Institute of Psychosocial, Palliative and End-of-Life Care Operations Committee and medical director of the Harold and Shirley Lederman Palliative Care Centre. Additional information on competing interests can be found in the CMAJ publication.
Tara Gomes reports research funding from the Ontario Ministry of Health, the Ontario College of Pharmacists, and the Canadian Agency for Drugs and Technologies in Health; consulting fees from the Auditor General of British Columbia; payment for expert testimony from the Office of the Chief Coroner of Ontario; and travel support from Indigenous Services Canada. She is a Tier 2 Canada Research Chair in Drug Policy Research & Evaluation.
Sarina Isenberg reports travel funding from the American Association of Hospice Palliative Medicine. She is director of the St. Joseph’s Villa Foundation Board of Directors and a member of the Temple Anshe Sholom Succession Planning Committee
Lau J, Scott M, Everett K, Gomes T, Tanuseputro P, Jennings S, Bagnarol R, Zimmermann C, Isenberg S. Association between opioid use disorder and palliative care: a cohort study using linked health administrative data in Ontario, Canada. CMAJ. 2024 April 29. doi: 10.1503/cmaj.231419.
A research team at Toronto General Hospital Research Institute (TGHRI) has built an artificial intelligence (AI) learning model to understand complex biological interactions from large-scale datasets of the analysis of single cells.
Recent advancements in the study of the genes and gene expression patterns in single cells have provided a wealth of data that enables researchers to learn about cellular diversity, function, and how cells respond to various conditions. The use of a technique called single-cell RNA sequencing—a method that measures the levels of gene expression in each cell to determine how it functions—has led to the development of comprehensive data atlases.
“The large volume of sequencing data has created huge analytical challenges,” says Dr. Bo Wang, Scientist at TGHRI and senior author of the study. “To address this, we wanted to develop a foundation model to employ machine learning to decode and predict single-cell behaviours from sequencing data.”
A foundation model can be described as a giant database of information that is trained on a large number of diverse datasets and can be adapted for a variety of tasks. Language models, like chatGPT, are trained on text to learn patterns and meanings in language. Then, the model can be used to assist with tasks such as answering questions, summarizing text, or translating languages.
“While texts are made up of words, cells can be characterized by genes and the protein products they encode,” says Haotian Cui, doctoral student in Dr. Wang’s lab and co-first author of the study. “Using this principle, we developed a foundation model called scGPT (singe cell GPT) to examine single cell biology by pre-training on over 33 million cells.”
By training on a diverse dataset containing millions of cells from different tissues and conditions (i.e., cell types from 51 organs or tissues and 441 studies), scGPT has learned to understand patterns in gene expression and cell behavior and has been taught to create new information based on what it learned. Its main part uses special tools called transformer blocks to help it understand and process the data. After its initial training, its settings can be adjusted to make it work better with new information, which can be useful for various tasks.
The team found that scGPT is effective for tasks such as identifying cell types, predicting gene activity in cells, correcting batch effect errors in sequencing data, and uncovering important gene interactions that vary depending on the cell type or condition. This approach enhances the modeling of single-cell sequencing data and provides valuable insights into gene-gene interactions specific to different conditions such as cell states and gene expression disruptions.
“The release of scGPT models and workflows will be able to accelerate research in cellular biology and beyond, offering a standardized approach for analyzing single-cell omics—the profiling of single cells in various populations,” says Chloe Wang, co-first author of the study and doctoral student at TGHRI.
By leveraging the power of a pre-trained generative AI model, researchers hope to pave the way for innovative therapeutic strategies and deepen understanding of cellular processes.
“For the future, our goal is to make our model smarter and better at understanding how cells work in different situations,” adds Dr. Wang who is also Chief AI Scientist at UHN and co-lead of the UHN AI Hub.
Since the preprint of this study in May 2023 and the release of scGPT, it has significantly impacted the field, with over 13,000 installations and 55 citations before its official publication.
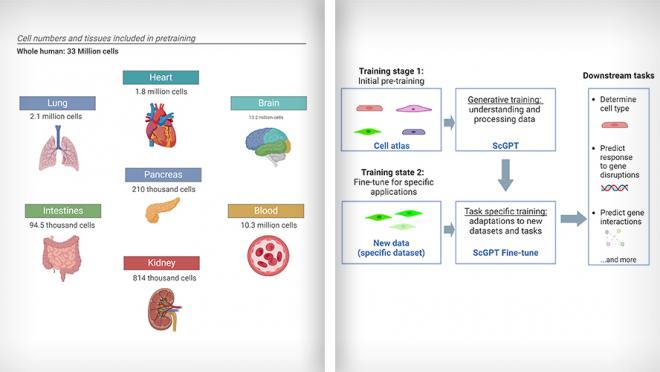
(L) scGPT was trained using single cell RNA-sequencing data from 33 million normal human cells from various organs. (R) Simplified workflow of scGPT. Images adapted from Cui et al., 2024, Nature Methods using BioRender.
This work was supported by funding from the Natural Sciences and Engineering Research Council of Canada, the Canadian Institute For Advanced Research AI Chairs Program, and the Peter Munk Cardiac Centre at the University Health Network.
Dr. Bo Wang is a Tier 2 Canada Research Chair in Artificial Intelligence for Medicine and an Assistant Professor at the University of Toronto.
Dr. Bo Wang is on the advisory board of Vevo Therapeutics and co-author Dr. Nan Duan is an employee of Microsoft and holds equity in the company.
Cui H, Wang C, Maan H, Pang K, Luo F, Duan N, Wang B. scGPT: toward building a foundation model for single-cell multi-omics using generative AI. Nat Methods. 2024 Feb 26. doi: 10.1038/s41592-024-02201-0. Epub ahead of print. PMID: 38409223.
Krembil’s annual Research Day is a unique opportunity to exchange ideas, foster collaboration and celebrate research achievements.
This year’s event, held at the MaRS Centre on Thursday, April 11, 2024, welcomed 230 trainee researchers, faculty and staff who showcased the breadth of talent within our Institute and our collective dedication to advancing scientific knowledge.
The event opened with a welcome video that shined a spotlight on what it is like to be a Krembil trainee, as well as opening remarks from Dr. Jaideep Bains, Director of the Krembil Research Institute. Dr. Bains commended trainees at all levels for their diverse talents, dedication to research and outstanding contributions to the Institute.
Throughout the event, nine trainees delivered oral presentations that highlighted achievements across the Institute’s three research pillars: brain and spine, vision, and bone and joints. The event also featured more than 70 poster presentations from trainees and research staff that facilitated scientific dialogue and idea sharing.
This year's Research Day also featured a special “Lunch and Learn” event. Internationally renowned communications expert Bri McWhorter, Founder and CEO of Activate to Captivate, shared valuable insights on science communication in a talk titled, “The Art of Delivering an Unforgettable Presentation.”
Trainees benefited from McWhorter's expertise in storytelling, learning strategies for creating connections with audiences, navigating key moments in talks and crafting engaging narratives.
The event concluded with a keynote address from Dr. Anne-Marie Malfait, Professor of Internal Medicine and The George W. Stuppy, MD, Chair of Arthritis at Rush University, titled “Chronic Joint Pain: Five Short Lessons from Osteoarthritis.”
Click here to watch a short slideshow recapping this year's Research Day.
“Thank you to everyone who joined us for our annual Research Day. Your participation made the event an inspiring celebration of the breadth of ideas and expertise at our Institute and the collaborative spirit that fuels our success,” said Dr. Bains. “I look forward to carrying this momentum forward as we plan for next year’s Research Day and more opportunities for our research community to come together.”
The Krembil community thanks the many individuals who made this year’s Research Day possible, including the Krembil Trainee Affairs Committee—chaired by Dr. Mary Pat McAndrews—the Krembil Communications and Administration teams, the oral presentation session Chairs—Drs. Joan Wither and Bill Hutchison—and everyone who served as judges for the oral and poster presentations.
Presentation Awards
The following trainees and postdoctoral researchers received awards for best oral and poster presentations:
Poster Presentations - Graduate Student Category:
● 1st place: Zi Xuan Zhang
● 2nd place (tied): Ain Kim
● 2nd place (tied): Anca Maglaviceanu
Poster Presentations - Postdoctoral Researcher Category:
● 1st place: Laura Whitall-Garcia
● 2nd place: Icaro Oliveira
● 3rd place: Cody Wilson-Konderka
Oral Presentations - Graduate Student Category:
● 1st place: Kabriya Thavaratnam
● 2nd place (tied): Brian Nghiem
● 2nd place (tied): Pedram Mouseli
Oral Presentations - Postdoctoral Researcher Category:
● 1st place: Emily Mills
A notable addition to this year's awards was the People's Choice Award for Oral Presentations, received by doctoral student Kabriya Thavaratnam for her exceptional research and presentation skills. Judges also awarded master’s student Nikou Kelardashti for her sex and gender-informed abstract, reflecting Krembil's commitment to promoting diversity and inclusion in research.
Congratulations to the winners and to everyone who presented their work. We look forward to seeing you at next year’s Research Day!
Research conducted at UHN's research institutes spans the full spectrum of diseases and disciplines, including cancer, cardiovascular sciences, transplantation, neural and sensory sciences, musculoskeletal health, rehabilitation sciences, and community and population health.
Learn more about our institutes by clicking below: